|
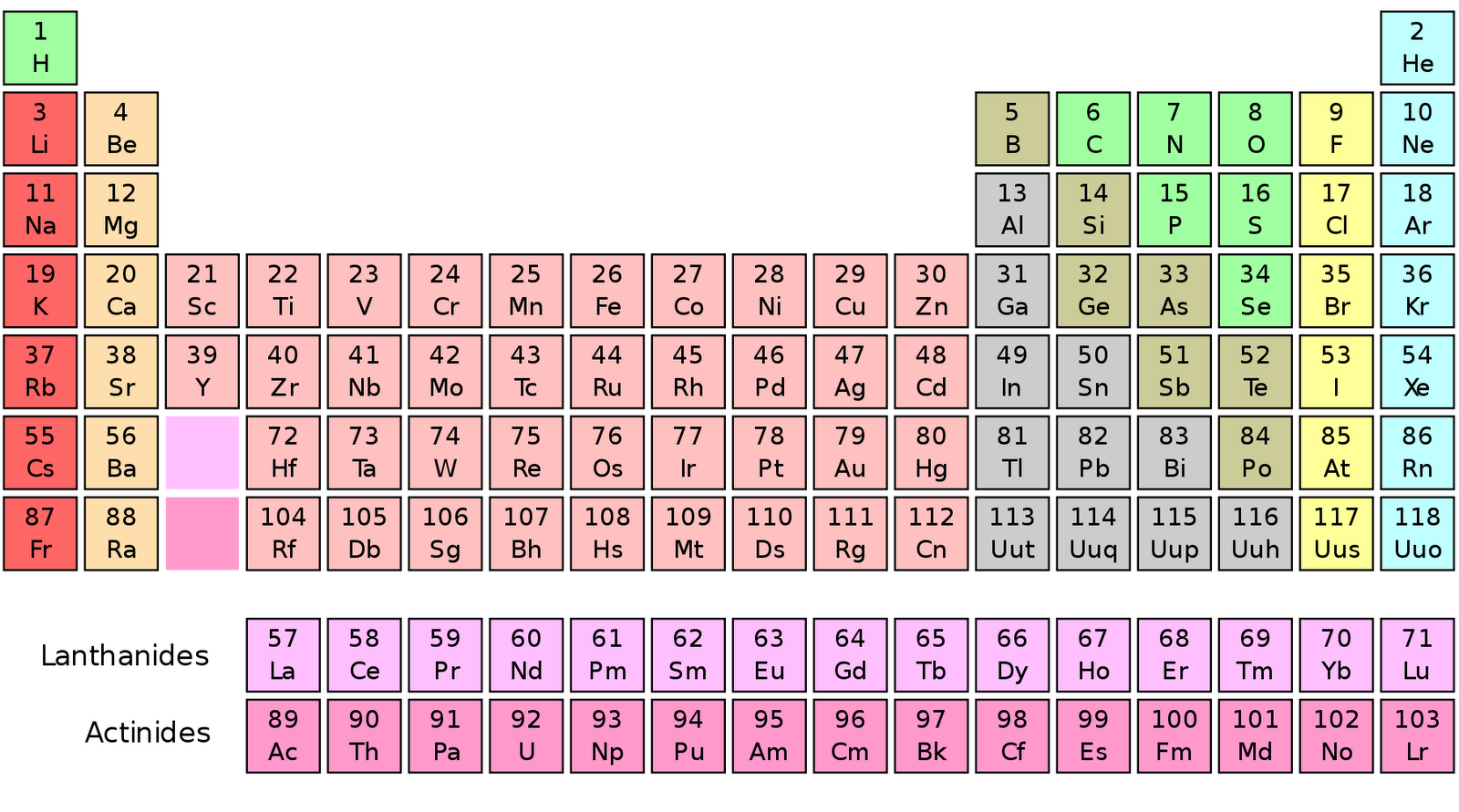
The total electrical charge of the nucleus is therefore +Ze. The number of electrons in an electrically-neutral atom is the same as the atomic number. Either way, we can find out the atomic number as the isotope symbol will consist of the chemical symbol.The atomic number or proton number is defined as the total number of protons in the nucleus and is given the symbol Z. For example, 14C or 14 6C are two ways to write the isotope of carbon. There are many ways in which we can write the isotope symbol. Last, if we know the isotope symbol of the element, we can find out the atomic number. It is easy to find out the atomic number of an element using a periodic table because the elements are present in order of increasing atomic number. It is a positive whole number denoted by letter z. You can see the atomic number of Al there. For example, say you want to know the atomic number of symbol Al (aluminium). Second, if we know the element name or symbol, we can find the atomic number using the periodic table. Because the number of protons is equal to the atomic number of an element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |





 RSS Feed
RSS Feed
